|
Dawn O’Connell, assistant secretary for preparedness and response at the Department of Health and Human Services, said in a Jan. The Biden administration has invested billions to increase production of rapid tests. There are three other at-home antigen tests that require a prescription and have FDA authorization. The Biden administration told us last month that eight of them are on the market. Thirteen fully at-home, over-the-counter antigen tests now have FDA authorization. 25, NIH announced it would spend $70 million to launch an Independent Test Assessment Program, an extension of RADx, to “ help identify manufacturers of high quality tests and encourage them to bring those tests to the U.S. The National Institutes of Health launched the Rapid Acceleration of Diagnostics (RADx) initiative in April 2020 with $1.5 billion in federal funding to speed the development of rapid COVID-19 testing. However, 2.2 million of those were subject to a recall because of “higher-than-acceptable false positive test results,” according to the FDA.īefore Biden took office, the FDA had authorized another at-home antigen test, but it required a prescription (the BinaxNOW COVID-19 Ag Card Home Test, authorized on Dec. “As of November 2021, Ellume had shipped approximately 3.5 million test kits to the U.S.,” spokesperson Annalyse Keller told us in an email. Also in mid-April, some CVS stores began selling another at-home test from Abbott that had received FDA authorization on March 31. 
The company received $30 million from the National Institutes of Health in 2020 to speed up development of its antigen tests, and in February, the Biden administration made a deal to pay $231.8 million for Ellume to manufacture the at-home tests and to help fund construction of a manufacturing plant in the U.S.Ī spokesperson for Ellume, a medical technology company headquartered in Australia, told us the tests were first available for sale to consumers at CVS Pharmacy, starting with some locations in Rhode Island and Massachusetts in mid-April and expanding to most CVS stores by the end of May. Both the Trump and Biden administrations have paid Ellume to develop and manufacture the test. The Ellume COVID-19 Home Test was the “first over-the-counter (OTC) fully at-home diagnostic test for COVID-19” authorized by the FDA, the agency’s press release in December said. What at-home antigen tests had been authorized by the FDA when Biden took office? Compared with previous variants, the omicron variant’s N protein is relatively unaltered, differing by a single short deletion and sometimes, one additional change. 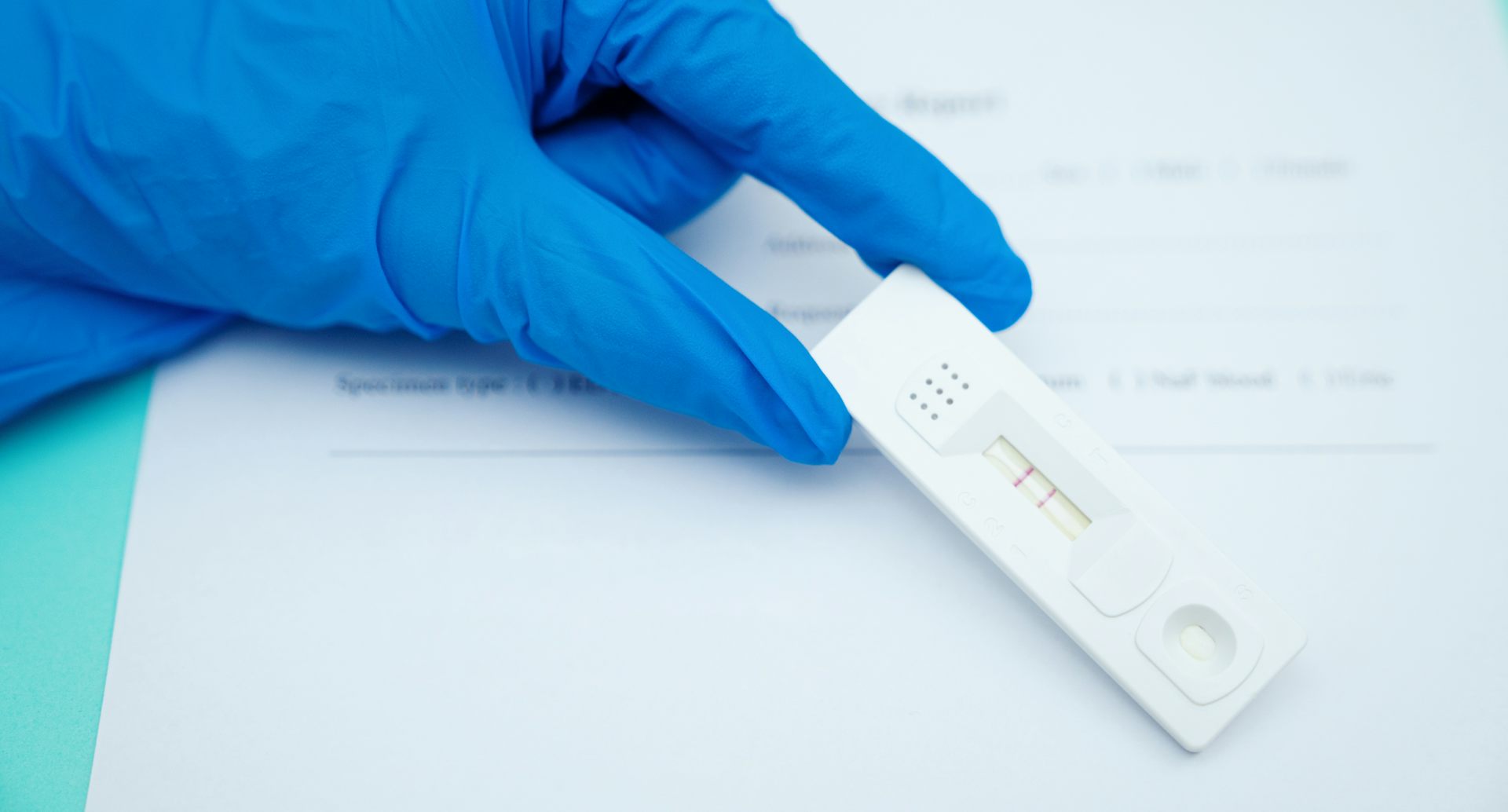
Indeed, in the case of the omicron variant, the majority of the mutations are in the gene that encodes the spike, or S, protein, which is the protein that is used in vaccines to prime the immune system and protect against disease. Typically, COVID-19 rapid tests use antibodies that recognize parts of the nucleocapsid, or N, protein of SARS-CoV-2 because more of that protein is made and because it’s less likely to change over time as the virus mutates. Sometimes known as lateral flow tests or lateral flow devices, rapid antigen tests for COVID-19 are similar to pregnancy tests in that they detect proteins in a specimen using antibodies embedded in a test strip. Some of these require a prescription, but the ones people are most familiar with are the self tests that can be purchased over-the-counter and can be performed entirely at home, with a person taking their own sample, running the test, and reading out the result within 15 to 30 minutes. What is a rapid antigen test?Īntigen tests are viral tests that check for the presence of SARS-CoV-2 viral proteins in a sample from a person’s nose or mouth to help determine if the person is currently infected with the coronavirus. President Joe Biden has said his administration will provide 1 billion free at-home tests to Americans, beginning Jan. We focus on the fully at-home antigen tests, which provide results in about 15 minutes. With that demand have come questions about the tests’ efficacy, how to use them and where to get them. 
The demand for at-home, rapid COVID-19 tests - sparked by the fast-spreading omicron variant in December - has continued in 2022.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
